Hygienic seals are critical components in industries where contamination control, cleanability, and regulatory compliance are non-negotiable. In food processing, pharmaceuticals, biotechnology, dairy production, and beverage manufacturing, sealing systems must prevent leakage while maintaining strict sanitary integrity. Unlike conventional industrial seals, hygienic seals are engineered specifically to eliminate microbial growth risks, withstand aggressive cleaning cycles, and comply with international sanitary standards.

1. What Are Hygienic Seals?
Hygienic seals are elastomeric or polymer sealing elements designed for use in sanitary equipment where product purity and contamination prevention are essential. They are commonly installed in:
- 펌프
- Valves
- Mixers
- Pipe connections
- Heat exchangers
- Filling systems
Their primary function is to maintain a leak-tight barrier without creating dead zones, crevices, or surfaces that could harbor bacteria.
2. Engineering Requirements for Hygienic Sealing
Hygienic sealing differs fundamentally from general industrial sealing due to several key engineering requirements.
Cleanability and Surface Integrity
Seals must withstand frequent Cleaning-In-Place (CIP) and Sterilization-In-Place (SIP) cycles involving:
- High temperatures
- Steam sterilization
- Chemical disinfectants
- Caustic and acidic cleaning agents
The seal design must avoid gaps, undercuts, or sharp transitions that could trap residue.
Material Safety and Compliance
Materials must comply with international regulatory standards, including:
- FDA (U.S. Food and Drug Administration) food-contact regulations
- EU Regulation (EC) No. 1935/2004
- USP Class VI (for pharmaceutical applications)
- 3-A Sanitary Standards
- EHEDG guidelines
These certifications ensure that seal materials do not leach harmful substances into the product stream.
Mechanical and Thermal Stability
Hygienic seals must maintain elasticity and compression set resistance under:
- Repeated thermal cycling
- High-pressure cleaning
- Dynamic loading in rotating or reciprocating systems
Failure in such conditions can lead to contamination, product recalls, and regulatory penalties.
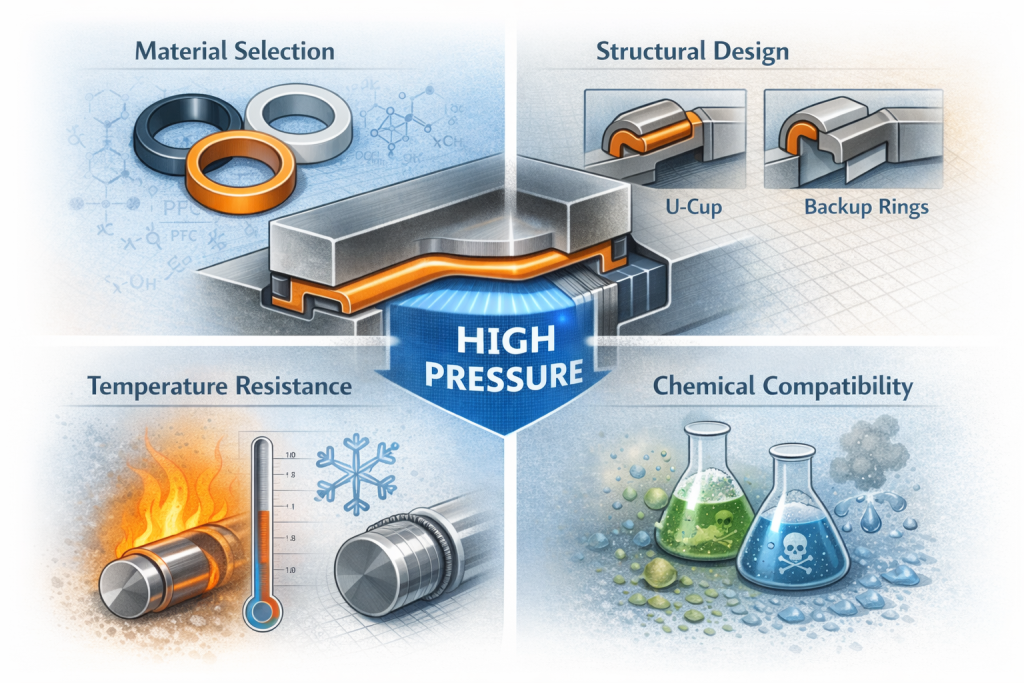
3. Common Materials Used in Hygienic Seals
Material selection directly impacts performance and compliance.
EPDM(에틸렌 프로필렌 디엔 모노머)
- Excellent resistance to hot water and steam
- Suitable for CIP/SIP processes
- Widely used in dairy and beverage industries
FKM(플루오로엘라스토머)
- High chemical resistance
- Suitable for aggressive cleaning agents
- 더 높은 온도 내성
Silicone (VMQ)
- Excellent flexibility
- Suitable for low-temperature applications
- Common in food and pharmaceutical systems
PTFE and Modified PTFE
- Exceptional chemical resistance
- 낮은 마찰
- Ideal for aggressive media
Each material must be selected based on temperature range, cleaning regime, media compatibility, and mechanical stress.
4. Hygienic Seal Design Considerations
Proper design is as important as material selection.
Compression Control
Excessive compression can lead to premature failure, while insufficient compression increases leakage risk. Controlled squeeze ratios are critical.
Surface Finish Compatibility
Contacting metal surfaces should meet sanitary surface finish requirements (commonly Ra ≤ 0.8 µm) to prevent microbial adhesion.
Avoidance of Dead Spaces
Seal geometry must prevent product entrapment. Hygienic groove designs eliminate crevices and support self-draining systems.
Dynamic vs. Static Use
Dynamic hygienic seals require additional consideration for wear resistance and friction management.
5. Failure Risks in Hygienic Sealing
When improperly designed or specified, hygienic seals may experience:
- Swelling due to chemical incompatibility
- Compression set leading to leakage
- Microbial ingress due to poor groove design
- Surface cracking after repeated sterilization cycles
Preventive validation testing, including chemical immersion testing and thermal aging analysis, is strongly recommended.
6. Lifecycle Cost and Risk Management
Although hygienic seals often carry higher upfront costs than general-purpose seals, they reduce:
- Contamination risks
- 생산 중단 시간
- Regulatory non-compliance penalties
- Product recall costs
Investing in certified hygienic sealing systems significantly lowers total lifecycle risk in sanitary industries.
결론
Hygienic seals are not simply upgraded industrial seals—they are precision-engineered components designed to protect product integrity, regulatory compliance, and operational reliability. Successful implementation requires careful material selection, validated design geometry, and strict adherence to sanitary standards.
Organizations operating in food, pharmaceutical, and biotech sectors should prioritize compliance-certified hygienic sealing solutions to ensure both safety and long-term cost efficiency.