In food processing and pharmaceutical manufacturing, maintaining product purity, process safety, and regulatory compliance is paramount. Hygienic seals are critical components that prevent contamination, maintain pressure integrity, and ensure the operational reliability of pumps, mixers, valves, and filling machines.
Selecting the right seal for sanitary applications is a complex task that requires understanding the interplay between material properties, seal geometry, system pressure, temperature, chemical exposure, and cleanability. Misjudged seal selection can lead to microbial contamination, product recalls, downtime, or accelerated wear.
Defining hygienic seals
Hygienic or sanitary seals are designed specifically for applications where cleanliness, sterility, and process safety are critical. They typically operate under low to moderate pressure, often in dynamic or semi-dynamic conditions, and are exposed to food ingredients, pharmaceutical solutions, CIP (Clean-in-Place) chemicals, and sterilization processes.
Two primary types dominate sanitary applications:
- Elastomeric seals: O-rings, lip seals, and gasketed inserts made from FDA-compliant elastomers such as EPDM, FKM, or FFKM.
- PTFE or polymeric seals: For applications requiring low friction, chemical resistance, or repeated sterilization cycles.
Seal design often integrates a metal or polymeric carrier to maintain shape and facilitate installation, particularly for dynamic pump or valve applications.
Material considerations
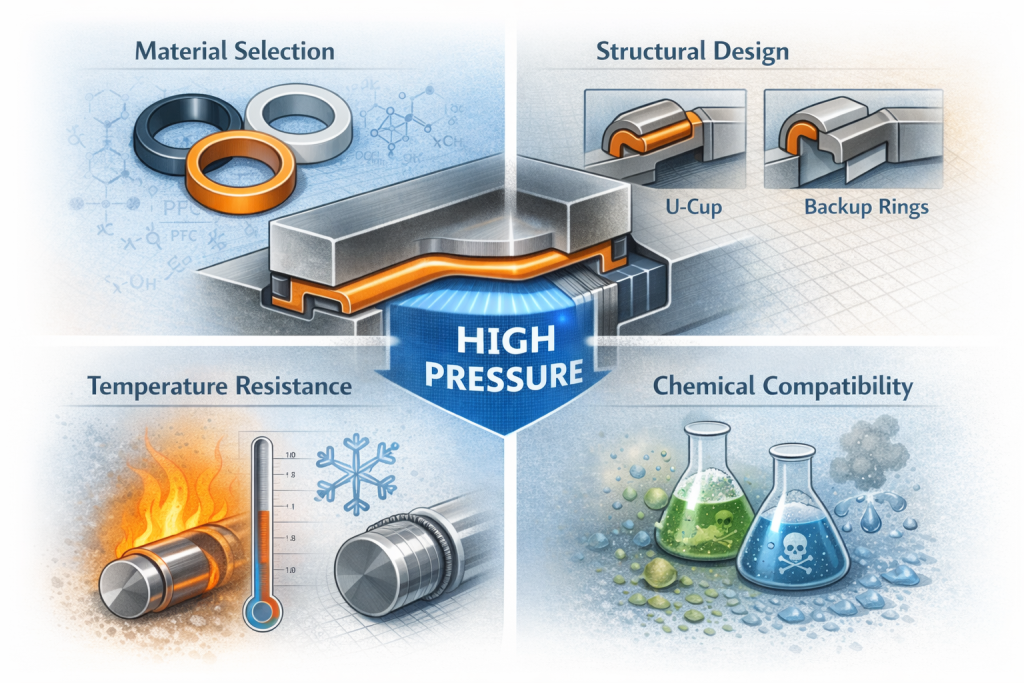
Material selection is the cornerstone of hygienic seal performance. Key criteria include:
- Regulatory compliance: Materials must meet FDA, USP Class VI, or EC 1935/2004 standards for direct contact with food or pharmaceuticals.
- Chemical resistance: Seals are exposed to cleaning agents such as caustic soda, peracetic acid, or hydrogen peroxide, requiring resistance to oxidation, hydrolysis, and swelling.
- Temperature stability: Steam sterilization or hot-fill processes demand materials that maintain elasticity and sealing force at elevated temperatures, often up to 150–180°C.
- Low extractables and leachables: In pharmaceutical applications, seal materials must minimize potential contamination from soluble compounds.
Common materials include EPDM for aqueous or steam applications, FKM for oil-based formulations, and FFKM for aggressive chemicals or extreme temperatures. PTFE and filled PTFE composites are often used for low-friction dynamic applications and sterile processes.
Seal geometry and system integration
Seal geometry is critical to performance in hygienic equipment. For static applications, gland design must provide uniform compression without creating crevices that can harbor microbes. Surface finish of mating parts must meet strict Ra and Rz targets to ensure proper sealing and cleanability.
Dynamic seals, such as pump mechanical seals, require precise tolerances, proper lubrication, and pressure balancing to maintain contact without excessive friction or wear. Spring-energized or pressure-assisted designs are common in high-speed mixers and rotary valves to maintain reliable sealing under varying process conditions.
Cleanability and sterilization
Hygienic seals must facilitate effective cleaning and sterilization. Crevice-free design, smooth surfaces, and flushable cavities reduce biofilm formation. Materials must withstand repeated CIP and SIP (Sterilize-in-Place) cycles without degradation.
Design strategies often include:
- Rounded corners and radii to eliminate dead zones.
- Integrated backup rings to prevent extrusion under CIP pressures.
- Surface coatings such as polished stainless steel or low-friction PTFE films to enhance cleanability and reduce adhesion.
Logique de sélection de l'ingénierie
Selecting a hygienic seal requires a systematic approach:
- Identify application type: Static flange gasket, rotary pump, valve stem, or piston seal.
- Define process conditions: Pressure, temperature, rotational speed, chemical exposure, and cleaning cycle.
- Determine regulatory and purity requirements: FDA/USP compliance, low extractables, and leachables.
- Match materials and geometry: Ensure chemical compatibility, thermal stability, low friction, and cleanability.
- Assess maintenance and lifecycle: Evaluate replacement intervals, ease of installation, and monitoring of wear or leakage.
Advanced engineering tools, such as finite element modeling of seal deformation and computational fluid dynamics to simulate flow past seal interfaces, can optimize performance and minimize contamination risks.
Case applications
In a pharmaceutical liquid-filling pump, a dynamic FFKM seal may be paired with a PTFE backup ring to handle steam sterilization and chemical CIP, ensuring zero leakage while maintaining low friction and long service life.
In food processing, such as dairy or beverage lines, EPDM or PTFE seals are integrated into flange gaskets and rotary valves with polished surfaces and crevice-free designs to prevent microbial growth during CIP cycles.
Conclusion
Hygienic seal selection in food and pharmaceutical equipment requires a multidisciplinary approach, combining materials science, mechanical design, surface engineering, and process understanding. The key considerations are regulatory compliance, chemical and thermal resistance, geometry optimization, and cleanability.
A well-designed hygienic seal ensures operational reliability, product safety, and process efficiency. By approaching seal selection systematically, engineers can minimize contamination risks, reduce downtime, and extend the lifecycle of critical equipment in demanding sanitary applications.